Sterile test isolators from Ortner ensure safe working conditions according to GMP Class A.
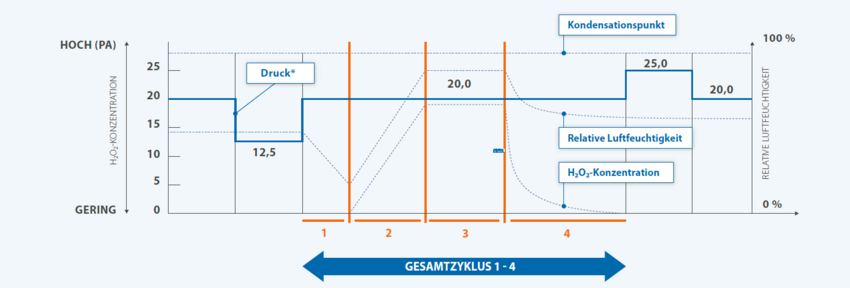
The integrated, fully automatic H₂O₂-decontamination around LOG6 germ reduction guarantees aseptic working conditions in the isolator.
Safe decontamination of the H₂O₂-before the start of the test ensures that sterility testing is carried out with the exclusion of false- negative results.































 Print out
Print out